This is an old revision of the document!
Table of Contents
Chronic Myeloid Leukemia
Introduction
History
Epidemiology
Hierarchy of Blood Cell Development - Hematopoiesis
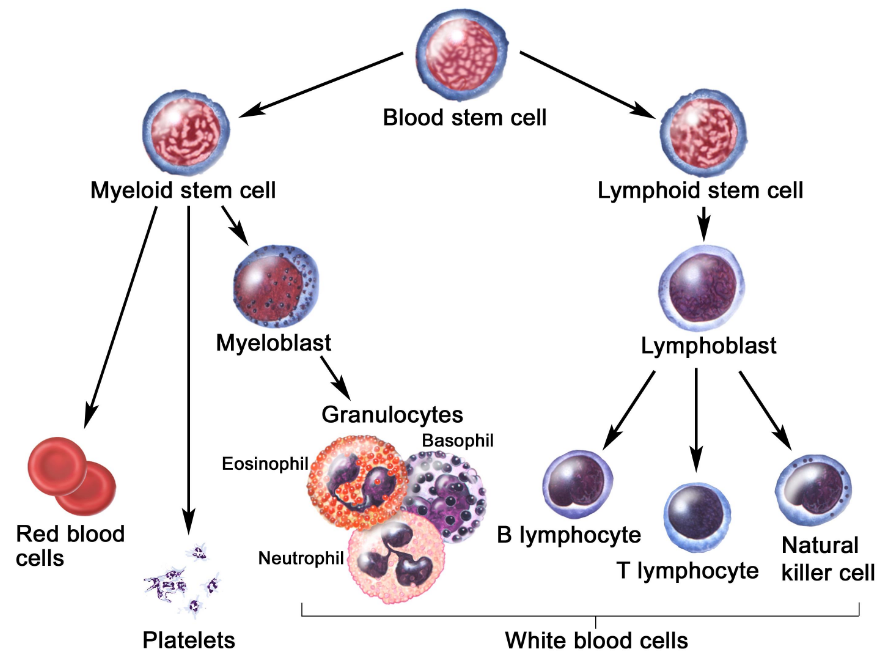
Blood cells develop in a committed, step-wise fashion. Blood cells are continually generated from self-renewing hematopoietic stem cells that give rise to non self-renewing progenitor cells. These progenitor cells are committed to give rise to cells that are more restricted in differentiation potential and finally to mature cells. This development is outlined in figure X at the right.
Hematopoietic stem cells (HSCs) are considered multipotent stem cells, which can differentiate into any blood cell type. Specifically, HSCs give rise to the lymphoid and myeloid lineages of blood cells. The myeloid stem cell gives rise to red blood cells, platelets, and granulocytes (i.e., neutrophils, basophils and eosinophils) while the lymphoid stem cells gives rise to B lymphocytes, T lymphocytes and Natural Killer Cells .
Leukemia
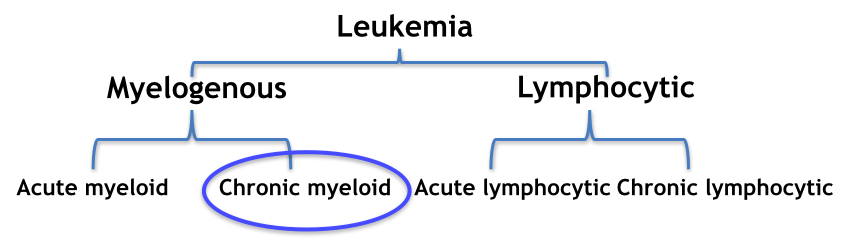
The abnormal presence of white blood cells in the blood is a direct translation of the word “leukemia”. In more general terms, leukemia can be characterized as a cancer of the blood that develops in the bone marrow (Canadian Cancer Society, 2017). There are many different types of leukemia, and they are grouped based on the type of blood stem cell they developed from (Canadian Cancer Society, 2017).
- Lymphocytic Leukemias: develop from abnormal lymphoid stem cells
- Myelogenous Leukemias: develop from abnormal myeloid stem cells
Moreover, the types of leukemia are further grouped based on how quickly the leukemia develops and grows. Acute leukemias start suddenly, developing within days or weeks, and chronic leukemias develop slowly over months or years. The four main types of leukemia are outlined in figure X below.
Figure X: Development of the four main types of leukemia
Signs and Symptoms
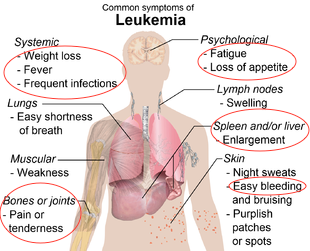
Symptom onset differs depending on the type of leukemia. In acute myeloid leukemia, symptoms develop rapidly, often initially presenting as flu (Sawyers, 1999). However, in chronic myeloid leukemia symptoms develop more slowly, and severity depends on the production levels of unregulated myeloid cells in the bone marrow. Due to the slow progression of the chronic phase, symptoms often develop slowly and diagnosis results from routine blood tests initially performed to diagnose other hypothesized diseases (Sawyers, 1999). In chronic myeloid leukemia, symptoms result from an increased white blood cell, specifically granulocytes. The typical symptoms include:
- Fatigue
- Due to a relative decrease in RBCs compared to WBCs. Relative reduction of RBCs results in symptoms of anemia resulting from low iron levels.
- Weight loss
- WBCs become localized in the spleen and liver, causing damage to these organs and resulting in enlargement of the liver and spleen.
- Easy bruising
- Due to the high production of WBCs (specifically granulocytes) which outcompetes the production of platelets (responsible for the blood clotting phenomenon).
- Frequent infections
- The overproduction of granulocytes are released into the blood from the bone marrow as immature cells that are incapable of their normal, pathogen fighting mechanisms.
- Bone pain
- Resulting from hyperplasia in the bones. The high turnover rate of myelogenous progenitors leads to pressure buildup in the bones.
Risk Factors
Diagnosis
Molecular Pathogenesis of Chronic Myeloid Leukemia
In 1960, Peter Nowell and David Hungerford, working in Philadelphia, described a consistent chromosomal abnormality in patients with CML (Druker, 2008). They consistently reported an acrocentric chromosome that was thought to be a chromosomal deletion (Druker, 2008). Through this discovery, Chronic Myeloid Leukmemia (CML) became known as the first human malignant disease to be linked to an acquired genetic abnormality (Kantarjian, Talpaz, Giles, O’Brien, & Cortes, 2006). The characteristic genetic abnormality of CML, the Philadelphia chromosome, is present in the bone marrow cells of more than 90% of all patients with CML (Kantarjian, Talpaz, Giles, O’Brien, & Cortes, 2006). The reciprocal chromosomal translocation between the long arms of chromosomes 9 and 22, as illustrated below in figure x, is the hallmark of CML (Kantarjian, Talpaz, Giles, O’Brien, & Cortes, 2006). This process fuses the Abelson murine Leukemia viral oncogene homolog (ABL) on chromosome 9 with the breakpoint cluster region (BCR) protein on chromosome 22, generating an oncogene that encodes the BCR-ABL protein, a constitutively active cytoplasmic form of the ABL kinase (Kantarjian, Talpaz, Giles, O’Brien, & Cortes, 2006). Interestingly, this reciprocal translocation occurs only in somatic cell lines as the sex chromosomes (germ lines) are not involved. Ultimately, this BCR-ABL fusion kinase leads to increased and unregulated growth of myeloid cells in the bone marrow and the accumulation of immature cells released in the blood.
The "Philadelphia Chromosome"
Normal Physiology of BCR Gene
The normal cellular BCR gene encodes a 160,000 dalton phosphoprotein associated with a serine/threonine kinase activity phosphorylating proteins containing the amino acids serine and threonine (Maru & Witte, 1991). Moreover, it activates GTPases such as RAC1 (cell growth/proliferation) and CDC42 (cell cycle progression).
Normal Physiology of ABL Gene
Non-oncogenic ABL is a member of the tyrosine kinase family (Warmuth, Danhauser-Riedl, & Hallek, 1999). It is normally regulated by auto-inhibitory mechanisms, involved in cell differentiation an division, and participates in many signalling pathways in the cytoplasm and the nucleus (Hantschel & Superti-Furga, 2004). It has been reported that the SH3 domain negatively regulates the activity of ABL, and the deletion of this domain turns ABL into proto-oncogene (Hantschel & Superti-Furga, 2004).
Figure X: The reciprocal translocation between chromosome 9 and 22, a hallmark of CML, generating the Philadelphia Chromosome BCR-ABL.
Mechanism of Action of the BCR-ABL Fusion Kinase
Several downstream signaling pathways are influenced by BCR-ABL. The fusion of BCR-ABL results in a constitutively activated intracellular tyrosine kinase, phsophorylating STAT5 transcription factor and inhibiting apoptosis (Fernandez-Luna, 2000).
Treatment
Gleevac (Imatinib)
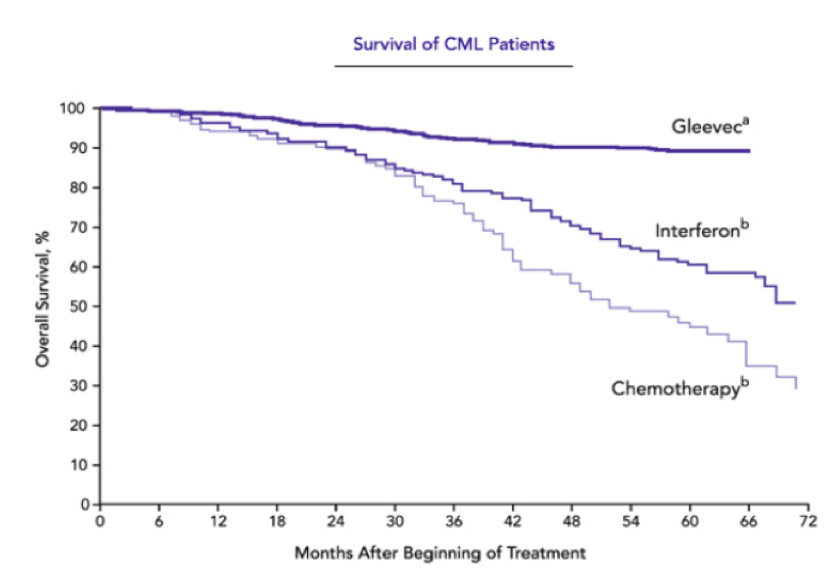
When Gleevec first came out it was called the miracle drug since it seemed as though it was able to cure chronic myeloid leukaemia (CML) due to its huge success rate. Gleevac works by binding to the closed/inactive conformer of BCR-ABL. This keeps the binding site for ATP closed and thus inhibiting the downstream signalling pathway which contributes to the progression of CML. During one of its first clinical trial conducted by Druker et al (2001), they looked at 54 patients with CML and prescribed them Gleevac of 300mg daily. Out of the 54 patients, 53 had a complete hematologic response within 4 weeks of therapy. A hematologic response is used to measure the recovery process of a CML patients by looking at whether their blood counts have returned back to normal and that there are no more immature cells in the blood. Druker et al (2006) did a 5 year follow up study and found that after 60 months on Gleevac, 98% of all their patients showed a complete hematologic response. The more outstanding result was that after 60 months the survival rates for patients were 89%. This is a huge improvement since only 30% of patients survived 5 years after being diagnosed with CML. Although within one year of the drug being released, some individuals started showing resistance to the drug due to CML cells having constant exposure to it. However, Gleevac still is the recommended and first line of drug for CML patients.
Dasatinib
After resistance grew on Gleevac, there was a development of another variation of the drug called Dasatinib. Unlike Gleevac, this drug targets the open/active conformer of BCR-ABL by binding to the active site and directly competing with ATP. During Clinical trials it showed it showed promising results. The researchers tested on 670 CML patients and broke them up into 4 groups, which were prescribed varying doses. At the end of their trials, 86-92% of patients saw complete hematologic response (Aguilera & Tsimberidou, 2009). There is research being done on combine drug therapy with Dasatinib and tyrosine kinase inhibitors. They have shown to work synergistically really well in producing growth inhibition and inducing apoptosis in CML cells (Aguilera & Tsimberidou, 2009). Dasatinib is currently used as a form of treatment for CML and is an effective therapy.
References
Aguilera, Dolly G, and Apostolia M Tsimberidou. “Dasatinib in Chronic Myeloid Leukemia: A Review.” Therapeutics and Clinical Risk Management 5 (2009): 281–89.
Druker, B. J., M. Talpaz, D. J. Resta, B. Peng, E. Buchdunger, J. M. Ford, N. B. Lydon, et al. “Efficacy and Safety of a Specific Inhibitor of the BCR-ABL Tyrosine Kinase in Chronic Myeloid Leukemia.” The New England Journal of Medicine 344, no. 14 (April 5, 2001): 1031–37. https://doi.org/10.1056/NEJM200104053441401.
Druker, Brian J., François Guilhot, Stephen G. O’Brien, Insa Gathmann, Hagop Kantarjian, Norbert Gattermann, Michael W.N. Deininger, et al. “Five-Year Follow-up of Patients Receiving Imatinib for Chronic Myeloid Leukemia.” New England Journal of Medicine 355, no. 23 (December 7, 2006): 2408–17. https://doi.org/10.1056/NEJMoa062867.
Druker, B. J. (2008). Translation of the Philadelphia chromosome into therapy for CML. Blood, 112(13), 4808–4817. https://doi.org/10.1182/blood-2008-07-077958
Kantarjian, H. M., Talpaz, M., Giles, F., O’Brien, S., & Cortes, J. (2006). New Insights into the Pathophysiology of Chronic Myeloid Leukemia and Imatinib Resistance. Annals of Internal Medicine, 145(12), 913. https://doi.org/10.7326/0003-4819-145-12-200612190-00008
Sawyers, C. L. (1999). Chronic myeloid leukemia. New England Journal of Medicine, 340(17), 1330-1340.
Warmuth, M., Danhauser-Riedl, S., & Hallek, M. (1999). Molecular pathogenesis of chronic myeloid leukemia: implications for new therapeutic strategies. Annals of Hematology, 78(2), 49–64. https://doi.org/10.1007/s002770050473