This is an old revision of the document!
Table of Contents
History
Chagas disease mainly affects people in Latin America. Chagas disease is an infectious disease which is caused by the protozoan flagellate Trypanosoma cruzi (T.cruzi) (Steverding, 2014). The earliest detection of T. cruzi, is from a Chinchorro mummy which was 9000 years old. The Chinchorro people were the first settlers of South America’s coastal region. During the 16th-19th century, several accounts of travellers and physicians with symptoms similar to Chagas disease have been recorded in a book published by Miguel Diaz Pimenta in 1707. Many years later, Carlos Chagas was informed about an insect that bit people by a railroad engineer worker. He then dissected the insects and found a large number of trypanosomes in their hindgut, which he then names T. cruzi. Dr. Chagas was able to link the insect to the trypanosomes and then to people’s symptoms (Steverding, 2014). Through his research, he discovered Chagas disease in 1909 (Moncayo, 2010).
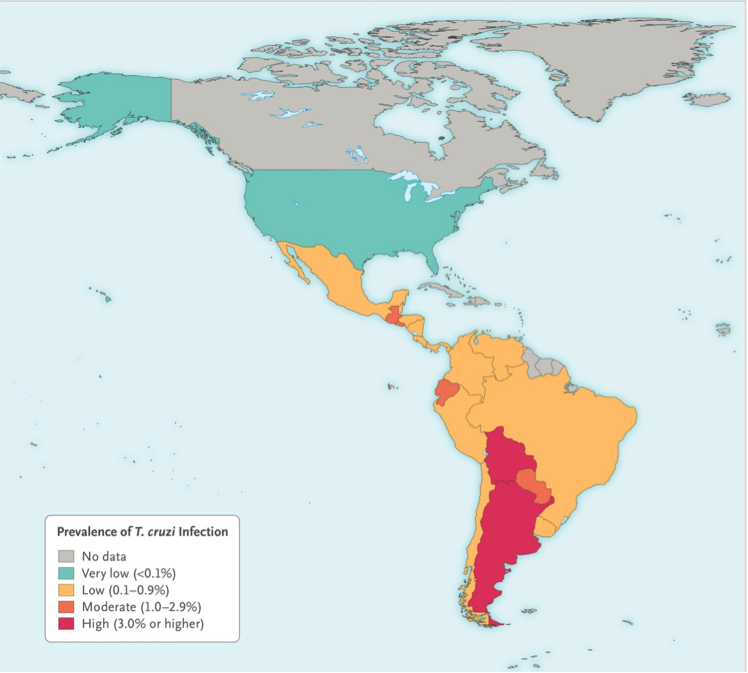
Chagas disease is predominantly in continental part of Latin America (World Health Organization, 2017). It has recently also been detected in the United States, Canada and many European countries due to travelling between these regions. It is estimated that Chagas disease infects 8 million people living in Latin American countries, 30-40% of which develop cardiomyopathy or digestive mega syndromes, or both (Rassi & Marin-Neto, 2010). Chagas disease is getting more attention as it is starting to become a global health concern since WHO recognized it to be the world’s 13th most neglected tropical disease in 2010 (Rassi & Marin-Neto, 2010). It has been estimated that more than 400,000 people have been infected with Chagas disease outside of Latin America with the US being the most affected (Steverding, 2014). T. cruzi infection rate is less than 1% per year, with the highest being 4% per year in hyperendemic Bolivian Chaco (Bern, 2015). It is encouraging for the scientific community to know that their efforts to decrease the occurrence rate has paid off since T. cruzi infection rate has decreased from 18 million in 1991 to 5.7 million in 2010(Bern, 2015).
Symptoms
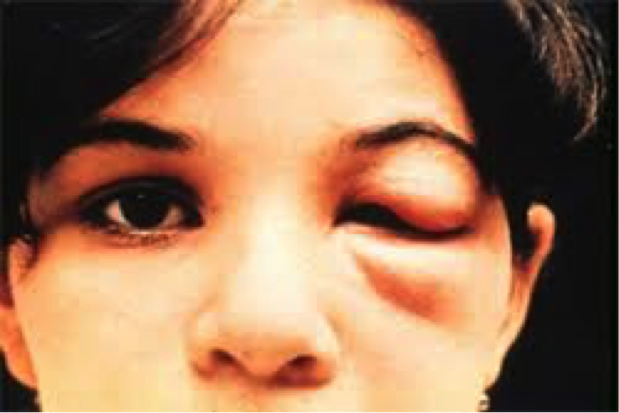
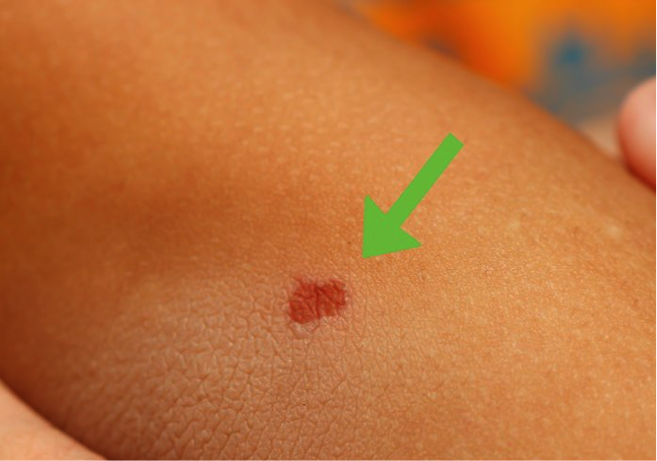
Symptoms of the Chagas disease vary between phases of the disease. About 95% of the cases with Chagas Disease do not show any symptoms in the acute phase (Teixeira, 2006). If the case does present any symptoms, they may show the following: Swelling or redness at the skin infection site
- Skin rash
- Swollen lymph nodes
- Fever
- Abdominal discomfort
- Headaches
- Fatigue
- Nausea, diarrhea, vomiting
- Muscle and joint pains
- Respiratory disturbances
- Cyanosis
- Coma
Patients who have above acute-phase symptoms, have them resolved in about 3-8 weeks spontaneously (Teixeira et al, 2006). If the person’s immune system is weakened, then occasionally the acute infections show chronic symptoms. Less than 5% of patients with the above presented symptoms die in the acute phase. The cause of death in the acute phase would be due to myocarditis or meningoencephalitis along with some complications like bronchopneumonia. After the acute phase, comes the intermediate phase or indeterminate phase, where the person shows no symptoms of the disease, according to most investigators and stays this way throughout their life. They may never know they even have Chagas disease if they showed no or mild symptoms in the acute phase. This phase lasts about 10-20 years before the person starts showing chronic symptoms. The disease, in the chronic stage, usually presents symptoms related to the heart in about 94.5% of all cases of Chagas disease (Teixeira et al., 2017). People with symptoms related to the heart are considered to have Chagas heart disease. These variations include heart insufficiency, and arrhythmias (Rassi, 2014). About 58% of the patients who die due to this disease have heart insufficiency and about 36.5% have been associated with unexpected deaths from arrhythmias. About 4.5% of the patients who have chronic Chagas disease present mega syndromes, which is a disease state that involves the esophagus, called megaesophagus, and the colon, megacolon. An important discovery to note is that the 58% of the patients who suffer from chronic Chagas heart disease, show signs of heart insufficiency and often die in between 7 months to 2 years after the onset of the symptoms (Teixeira et al., 2006). Other symptoms may include the following:
- Palpitations
- Fainting
- Cardiomyopathy
- Congestive heart failure
- Shortness of breath
- Emphysema
- Stroke
- Chronic abdominal pain
- Chronic constipation
- Difficulty swallowing
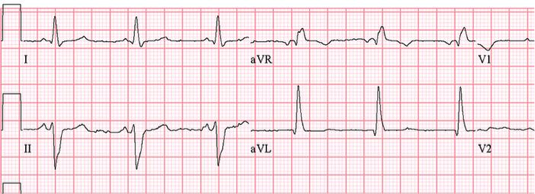
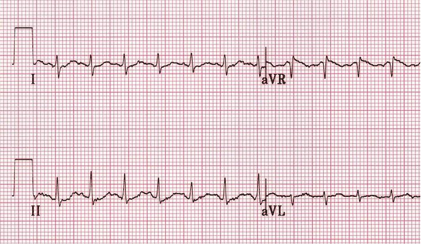
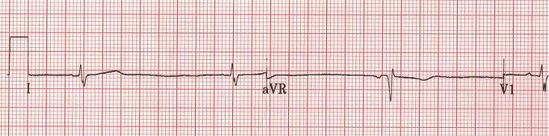
Clinical Presentation of Chronic Chagas Heart Disease
In Summary:
3 Stages of the lifelong Chagas’ Disease:
Acute Stage:
Symptoms can appear after 2-3 weeks after infection from the bug. This stage is common in children ages 10 or less. These symptoms include high fever, rashes, swollen nodes, and other ones mentioned previously. This stage lasts about 2-3 months in the patient’s life (Teixeira et al., 2006).
Indeterminate Stage:
Symptoms in this stage don’t exactly exist. This stage occurs mostly in people between ages 20-50 years and they may not even realize they have Chagas Disease! *but they can still pass it on* (Teixeira et al., 2006).
Chronic Stage:
Symptoms in this stage occur in about 40% of patients infected with T. Cruzi. The main and severe symptoms that characterize this stage is the aberrant function of the heart, the GI Tract, the brain. In short, more severe acute symptoms (Teixeira et al., 2006).
Risk Factors
Chagas disease originated in Latin America but due to low mobility in and out of these countries, the disease is not as predominant in North America. However, as travelling capabilities start to increase and more individual’s visit South America, the disease is beginning to spread north. The vectors prefer inhabiting the mud structures of villages and huts in latin america and are nocturnal. They come out at night to feed on human’s blood when they are sleeping.
Methods of Transmission
T. cruzi is transmitted through contact of faeces of infected insects of the Triatominae (kissing bugs or conenose bugs) subfamily. They are often called “kissing bugs” because they have the tendency to bite individual’s faces. When they bite, they also defecate, leaving their feces free to enter the bite site to transmit the parasite. Some of the most well known vectors of the disease include Triatoma infestans (kissing bugs), as well as Rhodnius proxlius and Panstrongylus megistus (Steverding, 2014).
Other methods of transmission in humans include:
- Transmitted congenitally from mother to baby
- Contaminated blood transfusions
- Contaminated organs from donors
- Contaminated food/drink (although this method of transmission is not common due to low pH levels in the stomach)
Life Cycle of T. cruzi
An infected vector is one that has trypomastigotes in their hindgut. Upon taking a blood meal from humans, they leave their feces on the skin and this is how trypomastigotes enter from kissing bugs to humans. Once they are in the human body, they lose their flagella and transform in the ideal. This is the ideal life cycle for them to reproduce exponentially within the cells. Once they have fully infected the cell, they transform back into trypomastigotes and burst out of the cell and enter the bloodstream. Once in the bloodstream, they can access other organs and once again transform into amastigotes inside the cell to replicate and infect the organ. Keep in mind that if a non-infected T.cruzi takes a blood meal from an infected human, they will become infected as well (Center of Disease Control and Prevention, 2015).
Mechanism
Recognition of Immune Cells Specifically Macrophages
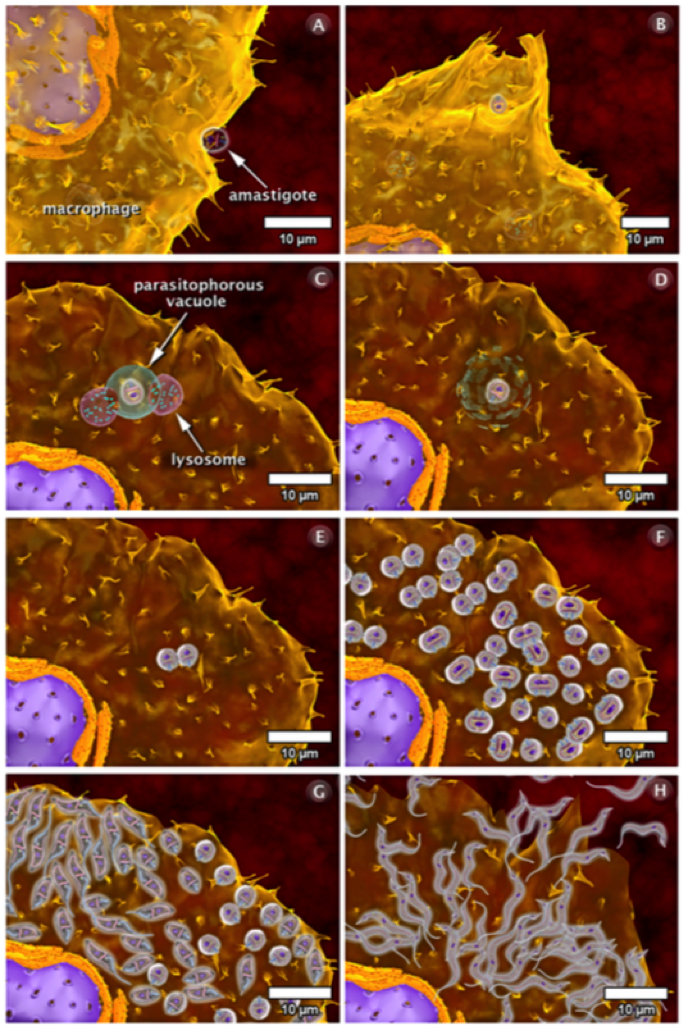
Once the infective trypomastigote enters the host, it binds to a complimentary immune cell such as a macrophage. The macrophages are recruited to the bite site because the parasite creates an open wound that requires a response from the immune system. The vector also releases histamine which causes the itchiness of the skin. The binding of the parasite to the macrophage initiates the internalization process using phagocytosis, forming pseudopods and the parasitophoros vacuole (Calvet et. al, 2012). In turn, this causes the recruitment of the host cell’s lysosomes to try and destroy the parasite. The lysosomes contain degrading enzymes. However, they are not successful. Once it is internalized, the trypomastigote transforms into the replicating amastigote and continues to multiply (Teixeira et. al, 2006). Once it has successfully completed cell division, it transforms back into the trypomastigote in order to initiate movement and infect the rest of the host cells. The parasite can only facilitate movement in the trypomastigote form. Eventually, the host cell will burst due to the increase in parasite density and the trypomastigotes gain access to the bloodstream and continues their infective routine to cells such as cardiomyocytes (Calvet et. al, 2012).
An amastigote can also bind to the surface of the macrophage and become internalized via phagocytosis (Teixeira, et., al, 2006). Using a similar technique, pseudopods and a parasitophorous vacuole are formed and fused with the lysosomes. The lysosomes digest the vacuole and the amastigote is free to divide rapidly (Calvet et. al, 2012). Again, the amastigotes transform back into the trypomastigote, causing the host cell to burst, and are then released into the bloodstream (Teixeira et. al, 2006).
Once T. Cruzi enters the host’s bloodstream, it needs to successfully recognize cardiomyocyte cells in order to infect the host cell. It does so using a variety of adhesion and internalization mechanisms. The trypomastigote binds to the cardiomyocyte cell in a similar way as the macrophage. Once it attaches to the surface, it forms the parasitophorous vacuole and transforms into the amastigote form to replicate. Following the rapid cell division, the parasite transforms back into the trypomastigote form and travels to the next cell to infect (Calvet, et. al, 2012).
There are multiple surface molecules on cardiomyocytes that allow for the internalization of T. Cruzi. These include carbohydrate residues such as glycoconjugates, specifically galactosyl, mannosyl, and sialyl. In addition, the mannose receptors on the surface are specifically inhibited by T.Cruzi, allowing the parasite to enter the cardiomyocyte. Extracellular matrix (ECM) components are also crucial in order for the parasite to recognize the cardiomyocyte. The parasite binds to a specific glycoprotein called fibronectin on the host cell. With the aid of it’s very own fibronectin receptor, it allows binding to the RGDS sequence (a series of amino acid residues) of the fibronectin and allows for entry into the cell (Calvet et. al, 2012). An third way T.Cruzi is internalized is using lipid membrane rafts. The parasitophorous vacuole on T. Cruzi binds strongly to the low density lipoprotein receptor on the cholesterol and sphingolipid areas of the raft, further allowing internalization (Calvet et. al, 2012).
Invasion/Internalization
Once T.Cruzi is successfully bound to the host cell, microfilaments are rearranged which encloses the parasite into the cell. The rearrangement in the cytoskeleton introduces a protrusion of the cardiomyocyte plasma membrane, creating a actin-based membrane skeleton to capture and internalize the parasite. This disturbs the structure of the cell, causing the breakdown of the cardiomyocyte fibres such as fibronectin (FN) and gap junctions that are needed to generate action potentials when the heart beats. As a result, this causes cardiac arrhythmias since the structure of the heart is compromised (Teixeira et. al, 2006).
A second way is through a lysosome dependent mechanism. The parasite causes an increase in cytosolic calcium that leads to depolymerization of the actin and allows the recruitment of lysosomes to the parasite binding site. The lysosomes allow entry into the cell. These elevated levels of calcium are caused by a sensitivity to leupeptin leading to a release of calcium from the sarcoplasmic reticulum calcium stores, and extracellular calcium influx through cellular membrane calcium channels (Teixeira et. al, 2006).
Pathophysiology
Once the parasite has successfully compromised the host cell, the host’s immune system starts generating a response in order to fight the infection. When the amastigotes start to divide rapidly they accumulate and damage the nerve cells and overall structure of the cell. They do so by causing necrosis of the myofibrils in the cytoskeleton and therefore, a dysfunction in the microtubule filaments, specifically fibronectin. Immune response cells are recruited to the site of damage but also end up accumulating and contributes to the enlargement of the cardiomyocytes. The cell will receive specific responses from cytokines, chemokines, and nitric oxide. Some of the genes involved in the accumulation include nitric oxide synthase 2, matrix metalloproteinase -2 (NMP-2), NMP-9, interleukin-6 (IL-6), and tumour necrosis factor-alpha. Although this is meant to be a defensive response against the parasite, it often results in cardiac fibrosis and hypertrophy due to the accumulation of cells (Chaves et al., 2016). Cardiac hypertrophy is the abnormal enlargement of the heart muscle due to an increase in cardiomyocyte cells and size (Calvet et al., 2012). Specifically, a build up of immune response cells, and the protrusion of the cardiomyocyte plasma membrane will cause the hypertrophy. Similarly, cardiac fibrosis is the abnormal thickening of the hearts valves characterised by a dense accumulation of collagen due to the over proliferation of cardiac fibroblasts that results from the disruption in the extracellular matrix (Calvet et al., 2012). As a result, this creates extremely flaccid heart muscle and causes the severe inability of the heart to contract properly. The heart will then attempt to overcompensate for this loss, and the left ventricle will become enlarged as an attempt to eject all of the blood out of the flaccid areas of muscle, leading to various cardiomyopathies and cardiac denervation (Calvet et al., 2012).
Furthermore, the disruption in the cytoskeleton deregulates the calcium influx which also a necessary component for efficient cardiac cell contractility. The figure below (label once all figures are in) shows the effect of infection on the cardiomyocyte architecture, representing cardiac fibrosis and hypertrophy. Image A represents an uninfected cardiomyocyte, in contrast with the infected cardiomyocyte, image B. In image B, there is an increase in the size of the cells, as well as the abnormal distribution of fibronectin (FN) labelled in green. As the cell became more infected, the FN labelling faded. In image C, FN is labelled red, and shows the the normal distribution of FN in cardiomyocytes. In contrast, image D shows the decrease of FN in infected cardiomyocytes. Lastly, image E represents the disorganization of myofibrils in infected cardiomyocytes. The arrows point to the T.Cruzi parasite. The disruption in the myofibril arrangement and calcium channels results in the inability to contract properly (Calvet et al., 2012). In summary, the constant destruction of cardiac fibres, the collection of collagen leading to fibrosis to replace destroyed myocytes and the hypertrophy of the remaining cardiomyocytes in order to compensate for the loss of myocytes all lead to heart failure and cardiac arrhythmias (Calvet et al., 2012).
Figure 17. These images are a comparison between an infected cardiomyocyte with an uninfected one. In image A, it is an uninfected cardiomyocyte. In image B, the arrow points to the T. Cruzi parasite, red represents enlarged cells, and the green represents disorganized fibronectin. Image C is an uninfected cardiomyocyte with the blue being the cell, and the red being organized and normally placed fibronectin. Image D displays enlarged cardiomyocytes in blue, with displaced and decreased amounts of fibronectin in red. The arrows point to the parasite T cruzi. Image E represents the invasion of the T. Cruzi parasite (the light blue circles where the arrow is pointing), and the green represents the disorganized and deteriorating microtubules necessary for heart contraction. (Calvet, et. al, 2012)
Treatment
The treatment overall focuses on killing the parasite and managing signs and symptoms. The history of Chagas treatment
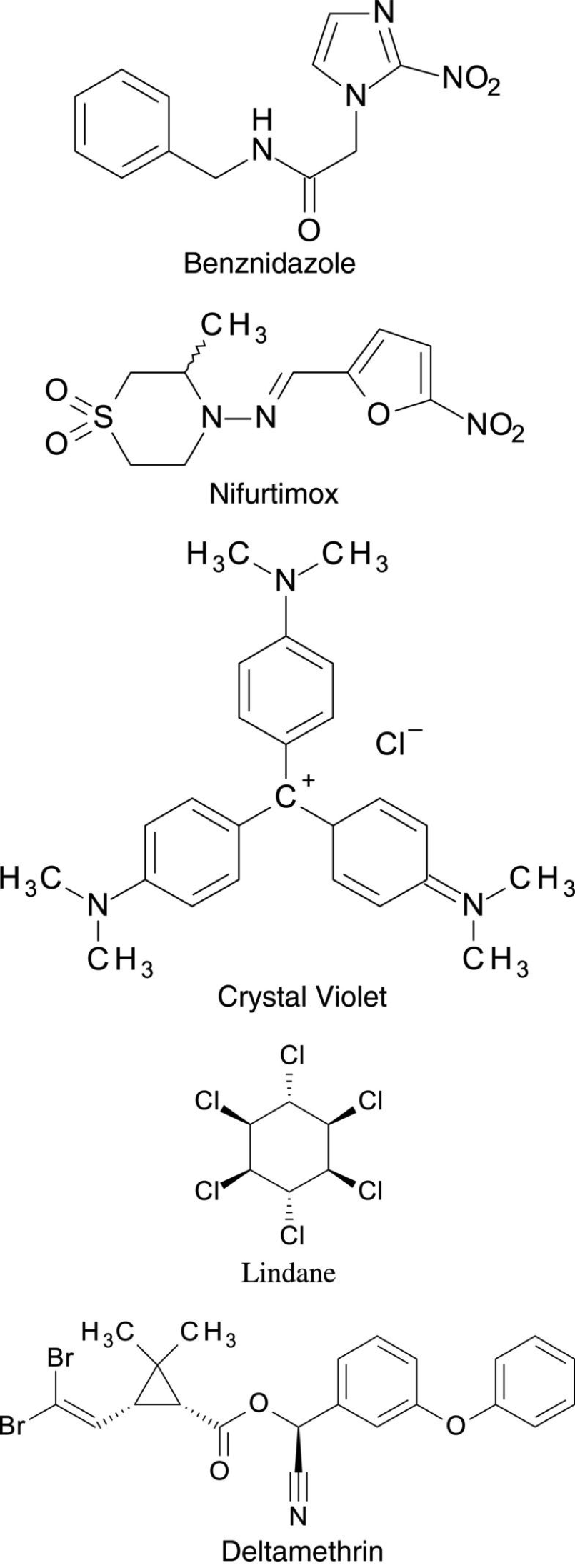
Testing for Chagas treatment began almost after its official discovery of this deadly infection. However, it took 50 years to develop two compounds into drugs in hopes to treat Chagas Disease. In 1966 Hoffmann-La Roche introduced Benznidazole. Soon after this discovery, another drug called Nifurtimox was also commercially available in the market as antichagasic medicine by Bayer in 1970. Moreover, In 1953 the dye crystal violet was also developed to kill T.cruzi parasite in blood preservations. The dye violet was used to protect blood used for transfusion from getting infected in blood banks in endemic areas to eliminate the disease as much as possible. Furthermore, Vector control started in the 1940s when the first organochlorine insecticides were introduced. Compounds such as Lindane and dieldrin were highly useful in killing the vector when sprayed over house walls. In the early 1980s, Synthetic pyrethroid insecticides like Deltamethrin and Cyfluthrin were also invented and they are very effective to control the domestic spread of the disease because they’re the most cost-effective and leave no unpleasant smell and marks on the treated walls.
Benznidazole (Benz-nidah-zol)
Benznidazole is a nitroimidazole (O₂NC₃H₂N₂H.) and was developed in 1966 and was officially released in 1972 at La Roche Laboratories. It is the primary treatment for the Chagas disease, and it’s FDA approved to use for children between 2 to 12 years. Nonetheless, patients are recommended to consume lower doses of it for their safety and adverse side effects. Through many clinical trials, it was found that Benznidazole is considered somehow effective in reducing the symptoms and detect the T.cruzi parasite inside the body. It was found that this treatment reduced 60 to 85 % of the acute cases, and in more than 90% of congenitally infected infants, if treated in their first year of life. However, the efficacy of Benznidazole in the chronic stage of the Chagas disease is still questionable. Many clinical types of research have shown that Benznidazole fails to treat Chagas patients in the chronical phase. Partially because the benefits of the drug in preventing cardiac and megacolon and megaesophagus manifestations are not yet clear (Gaspar et al., 2015).
The mechanism of Benznidazole against T.cruzi parasite requires the reduction of its nitro group by the parasite itself using the nitroreductases process. Through this process the drug will release free radicals that will react with proteins, lipids and DNA of the parasite and that will disturb the normal cell function of T.cruzi and its metabolism as well. Additional mechanisms involved in parasite killing by Benznidazole include T. cruzi NADH-fumarate reductase inhibition, phagocytosis improvement, and death by INF-γ. But, benzindolze is thought to be contributed to affect the human liver and that’s one of its adverse side effects (Gaspar et al., 2015).
Benznidazole as mentioned earlier it’s the only drug that’s approved by the FDA to treat Chagas disease, but it's considered to be very toxic and has severe side effects and the reason why is because it’s a nitroaromatic group which explains its significant toxicity. This toxicity is one of the main reasons of why Benznidazole isn’t considered an optimal drug to treat Chagas disease nevertheless it’s the only option to treat it. In addition, Benznidazole is prohibited for pregnant women, as many studies have shown that this drug can be crossed the placenta which will endanger the embryo with its potential of teratogenicity (Gaspar et al., 2015).
Common Side Effects
- Allergic dermatitis
- Anorexia and weight loss
- Insomnia
- Peripheral neuropathy
Nifurtimox (Ni-fur-ti-mix)
It is a Nitrofuran derivative (C10H13N3O5S) and is the only alternative to Benznidazole for the treatment of Chagas Disease. However it is not approved by the FDA. Moreover, Nifurtimox is used also to treat second stage African trypanosomiasis caused by a different parasite (Gaspar et al., 2015). The efficacy of this drug is similar to Benznidazole. However, Nifurtimox is more critical and causes more adverse effects. The mechanism of this drug is very similar to the mechanism of Benznidazole which is using the process of nitro reduction to produce nitro radicals that will affect the parasite on a cellular level. On the other hand, this drug uses Oxygen to produce oxygen species like superoxide and hydrogen peroxide ion that are also toxic to T.cruzi. This parasite is proved to be sensitive to oxidative stress as it has weak detoxification mechanisms due to the absence of catalase of peroxidase activity and reduced superoxide dismutase activity. Nifurtimox is not recommended for pregnant ladies for obvious reasons.
Common Side Effects
- Anorexia and weight loss
- Headache
- Polyneuropathy
- Nausea
- Vomiting
Patient Stories
Case Study 1
- Jose
- 46 years old
- California, USA
- Admitted to hospital with chest pain, shortness of breath
- Diagnosed with advanced heart failure
Jose and his wife were worried that he may have suffered a heart attack, due to his symptoms, but he was actually suffering from advanced heart failure at the young age of 46. Jose was put on the donor list, but in the meantime, one of his doctors at the hospital thought of a potential reason for this unpredictable case. She remembered learning about Chagas Disease in medical school, and how heart failure or death can be the result of this diagnosis. No doctor on the team has had a patient with a Chagas Disease diagnosis, so instead she called the Centers for Disease Control and Prevention’s physician hotline in order to get a second opinion. Based on Jose’s current status and medical history, they advised that Jose may indeed be suffering from Chagas Disease. The doctors ordered blood tests, which showed an infection with the parasite that causes Chagas Disease, Trypanosoma cruzi. Due to Jose’s advanced heart failure, treatment plans are bit more complicated. Jose needs a heart transplant, but the post-transplant medication could result in the parasite quickly dividing and spreading. Jose and his doctors would need to diligently monitor Jose’s health status for early signs of the infection worsening, in order to approve him for Chagas Disease treatment medication. Jose was lucky enough to receive a heart transplant and 2 months post-surgery, the Centers for Disease Control and Prevention conducted testing and detected early signs of his infection worsening. As a result, they provided Jose the treatment medications and his infection was then under control and his heart was functioning normally (Montgomery, 2017).
Case Study 2
- Maira
- 39 years old
- Kansas, USA
- Rejected blood donor
- No symptoms
Maira received a letter a few weeks after she donated blood for her workplace blood drive stating that her blood has been rejected. After leaving a message with the contact number, the Red Cross informed her that she has Chagas Disease. They provided her with a pamphlet on Chagas Disease, and she had the shocking realization that she got the parasite back when she grew up in a village in El Salvador with extreme poverty. Maira was referred to a specialist, but he said that Chagas Disease is untreatable. She was extremely angry and felt defeated, and decided to let it go for now, but Maira was weary everytime she had a health complication. A decade letter, Maira received a call from her sister who saw a news special about Chagas Disease, and identified a specialist that may be able to help. Maira contacted the specialist who started her on antiparasitic drugs. Although she wasn’t “cured”, her parasite counts have decreased, along with risks of serious complications (Loury, 2012).
References
- Steverding, D. (2014). The history of Chagas disease. Parasites & vectors, 7(1), 317.
- Moncayo, Á. (2010). Carlos Chagas: biographical sketch. Acta tropica, 115(1), 1-4.
- World Health Organization (2017). Chagas Disease. Retrieved from: http://www.who.int/mediacentre/factsheets/fs340/en/
- Rassi, A., & Marin-Neto, J. A. (2010). Chagas disease. The Lancet, 375(9723), 1388-1402.
- Bonney, K. M., & Engman, D. M. (2008). Chagas heart disease pathogenesis: one mechanism or many?. Current molecular medicine, 8(6), 510-518.*
- Chaves, A. T., Estanislau, J. D. A. S. G., Fiuza, J. A., Carvalho, A. T., Ferreira, K. S., Fares, R. C. G., … & da Costa Rocha, M. O. (2016). Immunoregulatory mechanisms in Chagas disease: modulation of apoptosis in T-cell mediated immune responses. BMC infectious diseases, 16(1), 191.
- Gaspar, L., B Moraes, C., H Freitas-Junior, L., Ferrari, S., Costantino, L., Paola Costi, M., … & Cordeiro-da-Silva, A. (2015). Current and future chemotherapy for Chagas disease. Current medicinal chemistry, 22(37), 4293-4312. Retrieved from https://research-repository.st-andrews.ac.uk/bitstream/handle/10023/9920/Gaspar_2016_Current_CurrMedChem_4293.pdf?sequence=1
- Steverding, D. (2014). The history of Chagas disease. Parasites & vectors, 7(1), 317. Retrieved from https://parasitesandvectors-biomedcentral-com.libaccess.lib.mcmaster.ca/articles/10.1186/1756-3305-7-317
- Calvet, C. M., Melo, T. G., Garzoni, L. R., Oliveira Jr, F. O., Neto, D. T. S., NSL, M., … & Pereira, M. C. (2012). Current understanding of the Trypanosoma cruzi-cardiomyocyte interaction. Frontiers in immunology, 3.
- Teixeira, D. E., Benchimol, M., Crepaldi, P. H., & de Souza, W. (2012). Interactive multimedia to teach the life cycle of Trypanosoma cruzi, the causative agent of Chagas disease. PLoS neglected tropical diseases, 6(8).
- Rassi Jr, A., Rassi, A., & Marin-Neto, J. A. (2009). Chagas heart disease: pathophysiologic mechanisms, prognostic factors and risk stratification. Memorias do Instituto Oswaldo Cruz, 104, 152-158.
- Teixeira, A.R.L., Nitz, N., Guimaro, M.C., Gomes, C., & Santos-Buch, C.A. (2006). Chagas disease. Postgrad Med J, 82 (974): 788-798.
- Rassi, A., (2014) Chagas’ Heart Disease. World Congress of Cardiology Scientific Sessions.
- Rassi, A., & Marin-Neto, J. A. (2010). Chagas disease. The Lancet, 375(9723), 1388-1402.
- Bern, C. (2015). Chagas’ disease. New England Journal of Medicine, 373(5), 456-466.
- Center of Disease Control and Prevention (2015). Chagas. Retrieved from https://www.cdc.gov/parasites/chagas/biology.html
- Montgomery, S. (2017). Heart Failure at Age 46? Centers for Disease Control and Prevention. Retrieved from https://blogs.cdc.gov/global/2017/04/14/heart-failure-at-age-46/
- Loury, E. (2012). Chagas' disease can cast a silent, lifelong shadow. Los Angeles Times. Retrieved from http://articles.latimes.com/2012/jul/08/local/la-me-chagas-20120708 - Ordered List Item